From Asia to North America: A Seamless Path to Commercial
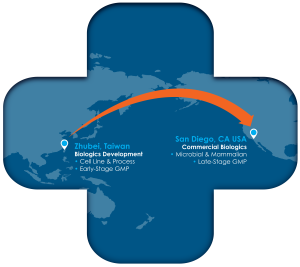 Biologics development has become increasingly global. Early-stage development and process optimization frequently occur in Asia, while late-stage clinical programs and commercial supply are often anchored in the United States. For sponsors navigating this transition, the challenge is not simply geographic — it is operational and regulatory.
Biologics development has become increasingly global. Early-stage development and process optimization frequently occur in Asia, while late-stage clinical programs and commercial supply are often anchored in the United States. For sponsors navigating this transition, the challenge is not simply geographic — it is operational and regulatory.
Moving a biologic program from an Asian development environment into U.S. commercial manufacturing requires more than tech transfer. It requires alignment in manufacturing control, regulatory expectations, and commercial discipline.
The Global Development Model Is Evolving
Asia has become a major engine for biologics innovation. Strong scientific infrastructure, cost efficiencies, and growing regional demand have accelerated early-phase development activity across the region. Cell line development, process optimization, and clinical-scale production are increasingly initiated outside the U.S.
However, as programs approach Phase III and BLA submission, sponsors often reassess long-term commercial strategy. The U.S. remains the largest biologics market globally and continues to set regulatory benchmarks that influence global approvals.
This creates a critical inflection point: transitioning from a development-focused environment to a U.S.-anchored commercial supply model.
The Risk Is Not Technical — It Is Structural
Most biologics processes are technically transferable. The greater challenge lies in structural alignment:
- Are quality systems aligned with U.S. regulatory expectations?
- Is documentation structured to withstand inspection readiness standards?
- Is process characterization sufficient to support long-term manufacturing control?
- Are supply chain systems built for sustained commercial distribution?
Without careful planning, gains achieved during early development can erode during late-stage transfer.
The U.S. regulatory environment increasingly emphasizes analytical rigor, validated control strategies, and sustained manufacturing performance. Sponsors must ensure that data packages, comparability strategies, and process controls are not only complete, but durable under inspection.
Designing for Commercialization from the Outset
A seamless path to commercial requires early integration of commercial principles:
- Establishing control strategies that anticipate scale-up
- Aligning documentation practices with BLA expectations
- Structuring tech transfer governance for traceability and accountability
- Conducting readiness assessments well in advance of PAI
This approach reduces variability during transfer and strengthens regulatory durability.
Commercial success is not achieved at approval — it is sustained through operational maturity and consistent manufacturing execution.
The Importance of U.S.-Based Commercial Infrastructure
For many sponsors, anchoring final commercial production in the United States offers several advantages:
- Direct alignment with FDA inspection frameworks
- Proximity to the largest biologics reimbursement market
- Reduced geopolitical and supply chain exposure
- Enhanced confidence among institutional buyers and healthcare systems
However, the transfer must be structured. A reactive approach — shifting manufacturing late in development without governance planning — introduces risk.
A disciplined transition model integrates:
- Early gap assessments
- Parallel validation planning
- Data integrity harmonization
- Defined comparability frameworks
When executed properly, geographic transition becomes a strategic advantage rather than a vulnerability.
A Connected Development-to-Commercial Strategy
The most resilient biologics programs treat global development and U.S. commercialization not as separate phases, but as a continuous pathway.
Sponsors benefit from partners that understand both environments — early-phase agility and commercial-stage discipline — and can bridge them without compromising manufacturing control.
As global biologics pipelines expand and regulatory frameworks continue to emphasize analytical robustness and operational consistency, seamless cross-regional execution will become increasingly important.
The question is no longer whether programs will move across regions.
The question is whether they will arrive commercially ready.
Now Operational: 2000L Single-Use Commercial Capacity in the U.S.
Bora Biologics’ expanded 2000L single-use capabilities support late-stage development and commercial GMP manufacturing in our San Diego FDA registered facility.